|
William Gaunitz, FWTS says hair bonding treatments can coat the hair to revitalize damage and breakage. “These products are restoring the microscopic bonds between the protein and molecules that make up your hair.” And since these damages can be caused by day-to-day factors like UV exposure, you may be surprised to find out exactly how damaged your hair may be at its core. Thankfully, these powerhouse bond repairing treatments help to, literally, rebuild and restore damaged strands, so you can have healthier, stronger hair without chopping it all off first. The hardest part is choosing which one you'll try. Here are the best bonding hair treatments for every hair type.The formation of a chemical bond involves a process to redistribute the electronic densities that originally belonged to different atoms. In connection with the fact that electrons on the outer level with the core are bound less firmly, they also play the main role in the formation of the connection. The number of bonds that is formed by an atom during a connection is called valency. Accordingly, electrons participating in the formation of a bond are called valence electrons. In the energy plan, there are atoms of different stability. The most stable is one with two or eight electrons at the outer level (the maximum number). In this case, this level will be considered complete. Such completed levels are characteristic of atoms contained in noble gases. In this connection, under ordinary conditions, they are in the state of a monatomic gas, chemically inert.įor atoms of other elements, incomplete levels are characteristic. When a chemical reaction occurs, the levels of the outer order are completed. This is due to the recoil or attachment of electrons, as well as through the formation of common pairs. 
The properties of atoms in this case are manifested in their striving to acquire a stable electronic shell of external significance. 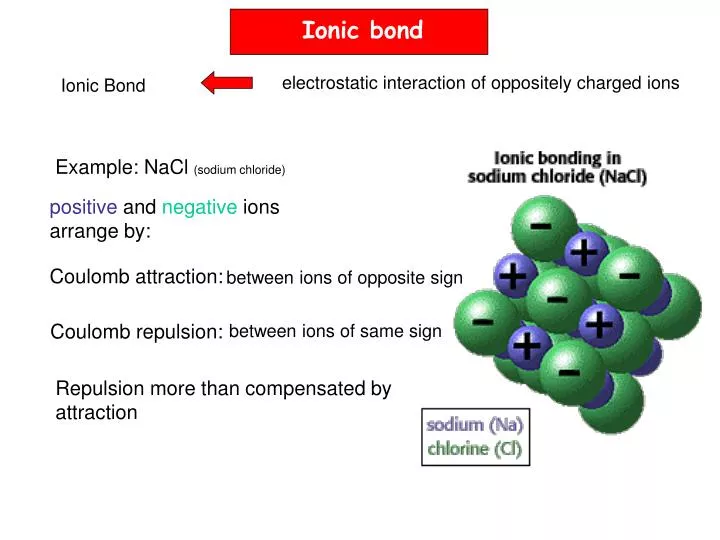
It will be either two-electron or eight-electron. This regularity is considered the basis of the theory of the origin of the chemical bond.įormation of the connection occurs accompanied by the allocation of a sufficiently large volume of energy. In other words, communication is formed exothermically. This is due to the fact that new particles (molecules) appear, which under normal conditions have greater stability.Įlectronegativity is one of the main features that indicate the nature of the emerging connection. This sign is manifested in the ability of atoms to attract electrons from other atoms.Įlectrostatic attraction between ions is an ionic chemical bond. Such an interaction is possible between atoms with a sharply differing electronegativity. Ionic bonding forms the appropriate ionic compounds. They consist of individual molecules exclusively in the vapor state. The ionic bond in the compounds of the crystalline (solid) state provides for the interaction of ions (negative and positive) located regularly. 
In this case there are no molecules.Ĭompounds for which an ionic bond is characteristic form the elements of the main subgroups of 1,2,6,7 groups.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
